Well, thanks for the terminology lesson, you may think, but so what? We already know that lipoic acid supplements provide health benefits. So does the fact that my lipoic acid is a “racemate” actually affect the value of the supplement? Does the presence of the S(-) form make any real difference?
You bet it does. The S(-)-form that’s taking up 50% of your supplement is not just a weaker cousin of the real thing, like the “other” isomers of alpha-tocopherol in the dl- product. In fact, S(-) lipoic acid is the R(+)-enantiomer’s “evil twin.” When he reported his findings about the opposing effects of the two forms of lipoic acid on the energy-producing powers of mitochondrial particles, for instance, Dr. Guido Zimmer stated that “The S(-)-enantiomer … part of the racemate, which is present as about a 50% impurity [our emphasis], needs to be eliminated.
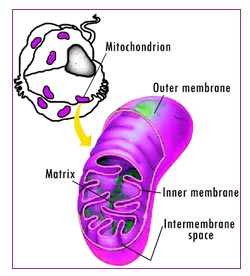 When you look at the differences between the R(+)- and S(-)- forms of lipoic acid in terms of their effects on the body’s metabolism of blood sugar, or their protective and antioxidant activities, or their effects on mitochondria.(the cellular “power plants” – see Figure 2), and the preliminary evidence on their effects on the aging process itself, we expect you’ll come to agree with Dr. Zimmer and other lipoic acid researchers (see sidebar: What Researchers Say). There are cases where the S(-)-enantiomer is merely less effective than R(+)-Lipoic Acid – and also cases where, at worst, the S(-)-form is just totally ineffective. But as you dig into the lipoic acid story, you’ll also see many cases in which the S(-)-enantiomer actually counteracts the enefits of R(+)-lipoic acid!
Learn More About What Researchers Say
When you look at the differences between the R(+)- and S(-)- forms of lipoic acid in terms of their effects on the body’s metabolism of blood sugar, or their protective and antioxidant activities, or their effects on mitochondria.(the cellular “power plants” – see Figure 2), and the preliminary evidence on their effects on the aging process itself, we expect you’ll come to agree with Dr. Zimmer and other lipoic acid researchers (see sidebar: What Researchers Say). There are cases where the S(-)-enantiomer is merely less effective than R(+)-Lipoic Acid – and also cases where, at worst, the S(-)-form is just totally ineffective. But as you dig into the lipoic acid story, you’ll also see many cases in which the S(-)-enantiomer actually counteracts the enefits of R(+)-lipoic acid!
Learn More About What Researchers Say
Learn More About What Researchers Say
Please consult with a health care professional before starting any supplementation program. The information contained on this site is general in nature and Company does not take any responsibility for any errors that may appear. Company has made every attempt to make the information as accurate as possible. However, Company does not warrant its accuracy. Please note that the statements on this web site have not been evaluated by the FDA.




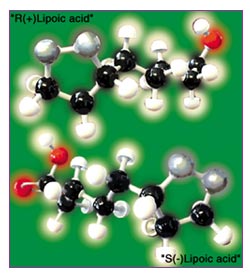 Of the two isomers of lipoic acid present in conventional supplements, the enantiomer which is identical to the one made by living things for their use – the natural form of lipoic acid – is the “R(+)-enantiomer,” or “ R(+)-lipoic acid.” “S(-)-lipoic acid” is a purely artificial molecule: it does not exist in nature but is produced as a byproduct in the normal method of producing commercial lipoic acid (see Figure 1).
Of the two isomers of lipoic acid present in conventional supplements, the enantiomer which is identical to the one made by living things for their use – the natural form of lipoic acid – is the “R(+)-enantiomer,” or “ R(+)-lipoic acid.” “S(-)-lipoic acid” is a purely artificial molecule: it does not exist in nature but is produced as a byproduct in the normal method of producing commercial lipoic acid (see Figure 1).


 When you look at the differences between the R(+)- and S(-)- forms of lipoic acid in terms of their effects on the body’s metabolism of blood sugar, or their protective and antioxidant activities, or their effects on mitochondria.(the cellular “power plants” – see Figure 2), and the preliminary evidence on their effects on the aging process itself, we expect you’ll come to agree with Dr. Zimmer and other lipoic acid researchers (see sidebar: What Researchers Say). There are cases where the S(-)-enantiomer is merely less effective than R(+)-Lipoic Acid – and also cases where, at worst, the S(-)-form is just totally ineffective. But as you dig into the lipoic acid story, you’ll also see many cases in which the S(-)-enantiomer actually counteracts the enefits of R(+)-lipoic acid!
Learn More About What Researchers Say
When you look at the differences between the R(+)- and S(-)- forms of lipoic acid in terms of their effects on the body’s metabolism of blood sugar, or their protective and antioxidant activities, or their effects on mitochondria.(the cellular “power plants” – see Figure 2), and the preliminary evidence on their effects on the aging process itself, we expect you’ll come to agree with Dr. Zimmer and other lipoic acid researchers (see sidebar: What Researchers Say). There are cases where the S(-)-enantiomer is merely less effective than R(+)-Lipoic Acid – and also cases where, at worst, the S(-)-form is just totally ineffective. But as you dig into the lipoic acid story, you’ll also see many cases in which the S(-)-enantiomer actually counteracts the enefits of R(+)-lipoic acid!
Learn More About What Researchers Say